|
12/28/2023 0 Comments N2f2 sigma and pi bonds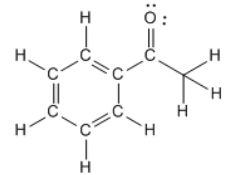
Here three pairs of electrons are being shared between two nitrogen atoms, so a triple bond is formed. Keeping all this in mind we draw the structure of $$ molecule. The octet of each atom should be complete. While drawing the structure ,consider only the valence shell electron i.e. Before drawing Lewis Dot Structure ,we need to keep in mind a few points in mind. The Lewis symbol of nitrogen will look like $:N \vdots $Ĭonsider the nitrogen molecule and draw its Lewis Dot Structure. According to the number of electrons being shared, calculate the number of bonds( bonding electrons) and lone pairs. Observe the number of electrons being shared between both the nitrogen atoms. These hybrid orbitals are allowed to overlap with ligand orbitals that can contribute electron sets for bonding.Hint: Draw the Lewis Dot Structure of the nitrogen molecule. Number of Orbitals and Types of HybridizationĪccording to VBT theory, the metal atom or ion under the influence of ligands can utilize its (n-1) d, ns, np, or ns, np, and orbitals for hybridization to yield a set of equivalent orbitals of definite geometry such as octahedral, tetrahedral, square planar and so on. It does not give a quantitative interpretation of the thermodynamic or kinetic stabilities of coordination substances.The theory presumes that electrons are localized in specific locations.No insight provided on the energies of the electrons.Failure to describe the tetravalency exhibited by carbon.The imperfections of the valence bond theory consist of: In HF, both the hydrogen and fluorine atoms share these electrons in a covalent bond. This bond forms from the overlap of the hydrogen orbital and the fluorine 2 p z orbital, which each have an unpaired electron. A covalent bond forms in between hydrogen and fluorine in hydrofluoric acid, HF. 
The F-F bond arises from overlapping pz orbitals, which each consist of a single unpaired electron.Ī similar situation takes place in hydrogen, H 2, but the bond lengths and strength are various between H 2 and F 2 particles. Fluorine atoms form single covalent bonds with each other. The diatomic fluorine molecule, F2, is an example. Valence bond theory can often explain how covalent bonds form. pi bonds are formed from sidewise overlapping whereas the overlapping along the axis containing the nuclei of the two atoms causes the development of sigma bonds. Sigma bonds and pi bonds differ in the pattern that the atomic orbitals overlap in, i.e.Covalent chemical bonds are directional and are also parallel to the area corresponding to the atomic orbitals that are overlapping.The paired electrons present in the valence shell do not take part in the formation of chemical bonds according to the valence bond theory. The presence of lots of unpaired electrons in the valence shell of an atom enables it to form multiple bonds with other atoms.The electron density in the location between the two bonding atoms increases as a result of this overlapping, consequently increasing the stability of the resulting molecule. Covalent bonds are formed when 2 valence orbitals (half-filled) coming from two different atoms overlap each other.The essential postulates of the valence bond theory are listed below. Examples of Sigma (σ) and Pi bonds (π)Īn example of Sigma bonds would be the simple C-H bond or C-X bond, while examples of pi bonds would be C =O and C ≡ N, where the first bond is a sigma bond, and the second/third bond are pi bonds. Double bonds consist of one σ and one π bond, while triple bonds consist of one σ and 2 π bonds. 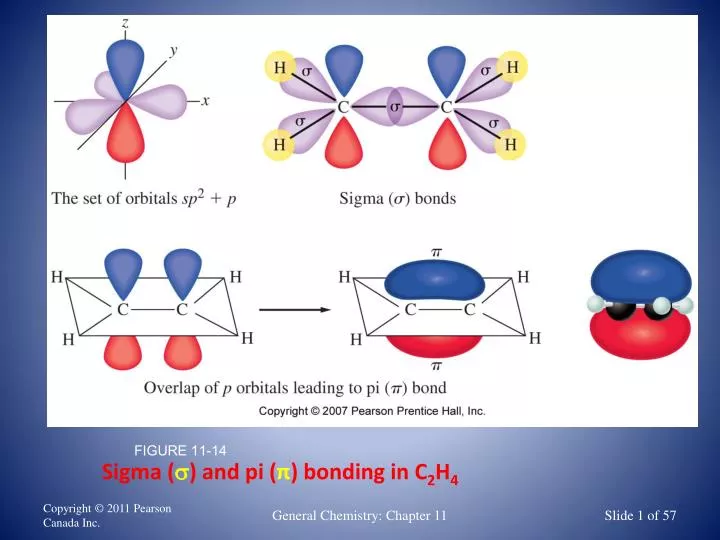
Single bond contains one sigma bond, double bonds have one sigma and one pi while triple covalent bond consists of one sigma and two pi bonds.īoth types of overlapping orbitals can be related to bond order. Pi bond formation takes place when there is overlap of two unhybridized p – orbitals. 
Sigma bond forms when orbitals overlap in the internuclear axis. VBT discusses the two types of overlapping orbitals. Larger the overlap, the more powerful the bond is. The bond strength depends on the overlapping of orbitals. It states that when half-filled orbitals of two atoms overlap, the electrons paired up as a result of this overlap. Valance Bond theory describes chemical bonding in determining the shapes of molecules. 9) Number of Orbitals and Types of Hybridization.5) Examples of Sigma (σ) and Pi bonds (π).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
